Project insights
My Role
UI Design
Design System
The Team
1 Designer, 1 Product manager, 4 Developers
Results
A note on how UX was done on this project
This engagement was originally scoped as a UI redesign — the client had a fully working system and needed it modernised. No formal user research, interviews, or commissioned usability studies were conducted. Instead, UX practice was woven into execution: heuristic evaluation of the legacy system, deep desk research into GxP regulations and pharma workflow standards, and workflow analysis through the product's own data models and screen flows.
This reflects a reality common in enterprise software agency work — UX doesn't always begin with a research phase. Sometimes you earn it screen by screen, and that's still worth documenting.
01 · INDUSTRY CONTEXT
Designing for a domain where compliance is not a feature
Life Sciences QMS software operates inside one of the most tightly regulated software environments in the world. Before touching a wireframe, the team had to understand the domain's language, its compliance obligations, and why every workflow step in the product exists — because most of them are legally mandated.
EU Regulation
EMA Annex 11
EU GMP Annex 11 governs computerised systems in pharma manufacturing. It mandates electronic record integrity, access controls, audit trails, and system validation. Any design change to a validated system requires documented change control.
US Regulation
21 CFR Part 11 (FDA)
FDA's Part 11 regulates electronic records and e-signatures in the US pharma sector. Systems must support ERES (Electronic Records/Electronic Signatures), full audit logging, and cannot allow silent deletion or modification of records.
International Standard
ICH Q10 — Pharma QS Model
ICH Q10 defines the pharmaceutical quality system model. OneQMS implements its four pillars — CAPA, change management, process performance monitoring, and management review — each needing its own UX treatment.
Why domain language shapes UX decisions
In regulated systems, terminology is not cosmetic — incorrect labels cause mis-triage, wrong approvals, and audit failures. The team built an internal domain glossary before designing any labels, status names, or navigation copy.
GxP
Good Practice Regulations
Umbrella term for quality guidelines (GMP, GCP, GLP, GDP). Any software used in GxP activities must be formally validated and comply with specific documentation requirements — which directly constrains how freely UX can change validated workflows.
CAPA
Corrective & Preventive Action
A structured investigation and action workflow for quality events. Consists of root cause analysis, correction, prevention, and effectiveness check. In OneQMS, CAPA is its own module under Quality — with multi-step lifecycle and assigned owners.
Change Control
Change Request (CR)
Any change to a validated process, document, or system must go through formal Change Control. The 8-stage workflow in OneQMS (Draft → Closed) is not a design choice — it maps to a regulatory requirement for traceability of change impact.
SOP
Standard Operating Procedure
A documented, approved instruction for how a regulated task is performed. SOPs must be version-controlled, reviewed on schedule, and distributed to relevant roles. Users must be trained on the current version — this ties Document Control directly to the Training module.
Deviation
Unplanned Departure from SOP
Any unintended departure from an approved procedure. Deviations are categorised by severity, investigated, and typically trigger a CAPA. In OneQMS this is a standalone Quality sub-module alongside Change Request and Audit.
Audit Trail
Immutable Activity Log
Every create/modify/delete action on a regulated record must be time-stamped and user-attributed in a tamper-proof log. This is a hard UX constraint — "delete" cannot silently remove records. This shaped how we designed destructive actions throughout OneQMS.
In pharma QMS design, ambiguity kills compliance. When a user can't tell if a status means "under review" or "rejected", they may skip a required step. That skip may fail an audit. That audit failure can cost hundreds of thousands in regulatory action.
02 · UX APPROACH
Embedded UX: making it work without a research budget
With no formal research phase commissioned, the team built a UX practice around what was available — the product itself, its regulatory context, and rapid iteration with domain feedback structured into critique cycles.
🧠 Domain Immersion
Client SME walkthroughs per module
Building internal glossary
Studied competitor systems: Veeva, ARISg
🔬 Heuristic Evaluation
Full audit of legacy system
Severity scoring per finding
Prioritised by regulatory risk impact
✏️ Design & Critique Cycles
Module-by-module wireframing
Weekly internal design critiques
Design system built in parallel
🧪 Dev-Cycle Feedback
Angular build reviews per sprint
UAT sessions as de-facto usability test
03 · QUALITY MODULE
Change Request & CAPA Workflows
The Quality module is the operational core of OneQMS. A single top navigation exposes seven sub-modules: Change Request, CAPA, Deviations, Audit, Incident, Quality Issues, and Customer Complaints. Each has its own multi-step lifecycle, assignee model, and document linkages — making navigation clarity and workflow visibility the primary UX concerns.
Change Request Lifecycle — 8 Mandatory Workflow Stages
Record shown: CLIN_SME_CR_QA_2023_00313 · Status: QA Approval · Group: No_Skip_Group — the status bar persists across all tabs: Change Request, Actions, Files, Reviewer/Approver, Effectiveness Check, Extension, Dependency, History.
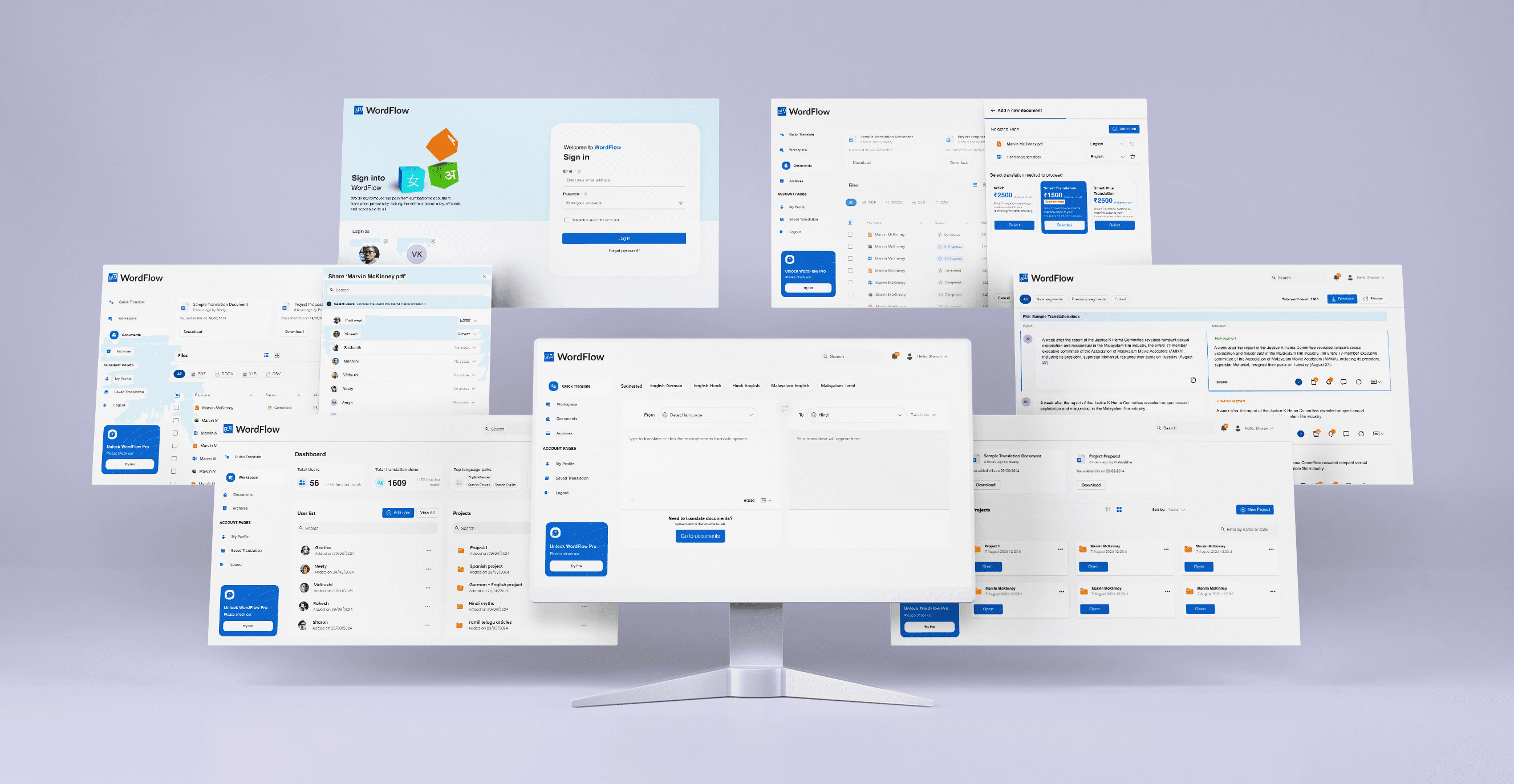
Quick Translate
Voice Input
Saving a translation
Saving a translation
04 · DOCUMENT CONTROL MODULE
SOP Library, Version Control & Issuance Tracking
Document Control is the backbone of GxP compliance. Every SOP, Work Instruction, Form, and Policy must be version-controlled, review-scheduled, approved, distributed, and linked to training obligations. Documents exist in multiple concurrent states — and the UI must make those states unambiguous, especially at audit time.
Quick Translate
Voice Input
Saving a translation
Saving a translation
05 · TRAINING & LMS MODULE
GxP Training Compliance, Gamification & Team Tracking
In a regulated environment, training is not professional development — it is a documented compliance requirement tied to job roles, SOP versions, and audit readiness. Lapsed training triggers system-level access restrictions. The Training module had to simultaneously serve individual learners, team managers, training administrators, and auditors.
Quick Translate
Voice Input
Saving a translation
Saving a translation
06 · DESIGN SYSTEM
Given OneQMS's breadth — 7 quality sub-modules, 4 document sub-modules, 5 training sub-modules — building a consistent foundation upfront was the only viable strategy. The design system was established in Figma before individual screen design began, and delivered to Angular development as a Storybook component library.
Quick Translate
Voice Input
Saving a translation
Saving a translation
06 · DESIGN SYSTEM
Compliance, efficiency & usability — measured
Outcomes tracked across 6 months post-launch across 140+ daily active users at 3 pharma client sites, covering the Quality, Documents, and Training modules.
With our requirements clarified through user stories, we restructured the information architecture to better align with user needs.
This content is protected.
To view, please enter password.




